 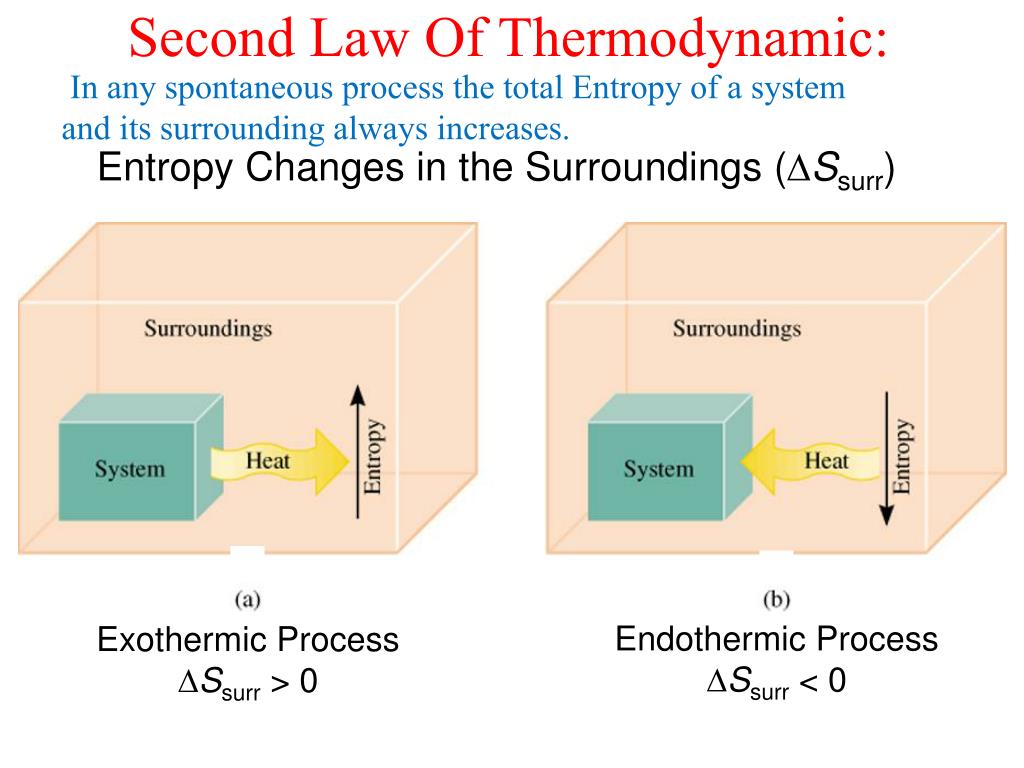 
(D)ΔG°reaction = ΔH°reaction – TΔS° reaction ΔG°reaction = ΔH°reaction – TΔS° reaction ΔG°reaction = (ΔG°f,products) - (ΔG°t,reactants) Reaction spontaneous at 25☌? (D) Is it spontaneous at 2000K? Using the standard free energy of formation in the equation Just like other thermodynamic functions, the free energy change for a reaction can be calculated by The GIBBS FREE ENERGY CHANGE can be determined from the GIBBS EQUATIONīy using change in enthalpy and change in entropy at constant temperature and pressure as shownįree energy and Spontaneity of a Reaction GIBBS FREE ENERGY, ENTHALPY, AND ENTROPY.The sign ΔG is used as the basis to determine whether a It is determined from difference in values of two The amount of energy that can be obtained from it. Is a measure of the spontaneity of a process and of A criterion for spontaneity that applies.Or you'll need to look them up in a table.)įormula: ΔS°reaction = ΔS°products - ΔS°reactants (Note, in this type of problem you'll either be given the molar entropy values of the reactants and products Reaction using standard absolute entropies instead ofĬalculate the change in entropy at standard state and 25☌ that accompanies the reaction:Ģ. The exact entropy change for the process can beĭetermined in the same way as the enthalpy change for the.Large increase in entropy resulting to a net increase in Of the surroundings of the system is decreased, the largeĪmount of heat released to the surroundings causes The heat released by the reaction increases the disorder Than the decrease of the surroundings, then the totalĬhange in disorder can be positive and the reaction That the change in disorder of the system is greater The disorder within the system increases sufficiently so THIS EXPLAINS WHY EXOTHERMIC AND ENDOTHERMIC REACTIONS MAY BE SPONTANEOUS.(IT MUST BE A POSITIVE VALUE – which corresponds to an ΔSuniverse = ΔSsystem+ ΔSsurroundings ≥ 0Įntropy change of the UNIVERSE is determined from the sum of entropy change of the 
The second law of thermodynamics states that the total entropy of the universeĪLWAYS INCREASES FOR A SPONTANEOUS PROCESS.One Mole of water is vaporized at 100☌ with the absorption of 40.7 kJ of heat.136)ĭetermine if the entropy change will be positive or negative for the following reactions:ġ) (NH4)2Cr2O7(s) → Cr2O3(s) + 4 H2O(l) + CO2(g) Sample Problem: Predict whether the entropy or disorderliness of the system where theįollowing changes occur is expected to increase or decrease.Type of process which does not need the.CHEMICAL THERMODYNAMICS (SPONTANEITY AND ENTROPY) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
